|
In other words, it should exhibit periodicity. Groups 1 and 17 on the table are the most reactive elements, while Group 18 (the Noble. The items in vertical columns (groups) must be similar in some manner and have some variation and gradual change as you move up or down the column. The reactivity of a substance can often be predicted through that elements location on the periodic table. These atoms are often called the alkali and alkaline earth elements. For example, potassium has a configuration Ar4s 1. The activity series is a list of elements in decreasing order of their reactivity. 1 ), the configuration is a closed shell of core electrons, plus s electrons in a new shell. 
The largest atomic radius of elements in their period. For atoms found in the first two columns of the periodic table (figure 1.6.1 1.6. The periodic table is being read like a book: Elements are listed starting from the top, far left element (hydrogen) across the whole row before. The repeating properties in each row of the periodic table, as observed by Mendeleev and others, reflect the repeating electron configurations in subsequent rows. Highly reactive, with reactivity increasing moving down the group. You do not have to include any Transition elements. List the first 12 elements mentioned in the song. The elements with properties intermediate between those of metals and nonmetals are called metalloids (or semi-metals). It must contain 4 periods and 8 families of a representative (short) periodic table. Non-metallic tendency increases going from left to right across the periodic table. Most periodic tables provide additional d ata (such as atomic mass) i n a box that contains each element’s symbol. A modern version is shown in Figure 4.6.1 4.6. Metallic tendency increases going down a group. The periodic table is one of the cornerstones of chemistry because it organizes all of the known elements on the basis of their chemical properties. 
Non-metallic character relates to the tendency to accept electrons during chemical reactions. This could be used to follow up some work on the periodic table where the trends in reactivity in groups 1 and 7 have been identified. 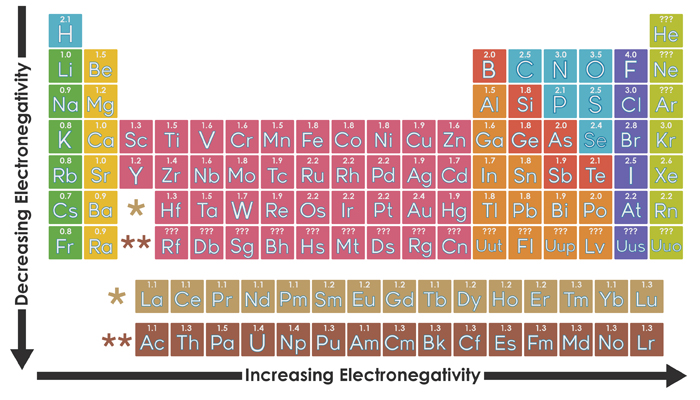
\)) acid has very weak bleaching characteristic.\), metals occupy the left three-fourths of the periodic table, while nonmetals (except for hydrogen) are clustered in the upper right-hand corner of the periodic table. Metallic character refers to the level of reactivity of a metal. Major periodic trends include: electronegativity, ionization energy, electron affinity, atomic radius, melting point, and metallic character. Chemistry for the gifted and talented: trends in reactivity in the periodic table.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
